Abstract
Introduction. Treatment of relapsed-refractory (r/r) diffuse large B-cell lymphoma (DLBCL) and high-grade B-cell lymphoma (HGBCL) has changed substantially in recent years, due to the incorporation (in a commercial setting or in the context of clinical trials) of new therapeutic options such as new monoclonal antibodies (MA), antibody-drug conjugates (ADC), bispecific antibodies (BA) or CAR-T cell therapy. The main objective of our study was to evaluate the frequency of use of these new therapies (NT) in Spain, and their impact on overall survival (OS).
Methods. This is a multicenter retrospective study. Patients with DLBCL and HGBCL were identified from the RELINF platform, where GELTAMO investigators register all patients with lymphoma diagnosed in each center from January 2014. Finally, 15 centers (2 out of 15 CAR-T cell providers) accepted to participate in the study, who were asked to complete additional information related with relapse and NT. Patients were divided in 2 cohorts according to date of documentation of relapse / refractory disease: 2014-2017 (first period) and 2018-2021 (second period). The use of NT in both periods and their impact on OS were analyzed.
Results We included 2162 patients in the analysis, 1891 DLBCL (51% germinal centre B-cell phenotype, 41% activated B-cell phenotype, 8% indeterminate), and 271 HGBCL (45% of them double/triple hit). Median age was 69 y/o, 72% were ≥ 70 y/o at diagnosis, and 49.8% were female. Four hundred and ninety four patients had relapsed or refractory disease, 183 in the 1st period, and 311 in the 2nd period. Median number of lines for r/r patients was 3 (2-7). NT were used in 353 patients, 30 in the 1st period and 111 in the 2nd period. New MA were used in 88 (28%) patients, being polatuzumab vedotin the most frequently used antibody (47, 54%) (tafasitamab 7, loncastuximab 5, brentuximab 11, others 10), and BA in 28 (9%), all of them within clinical trials. Twenty patients received NT in 1st relapse, 14 MA and 7 BA. From 211 patients who received > 2 lines of treatment, 128 were in the 2nd period, and 60 (47%) of them received CAR-T cell therapy, whereas 39 (30%) of these patients did not receive any NT.
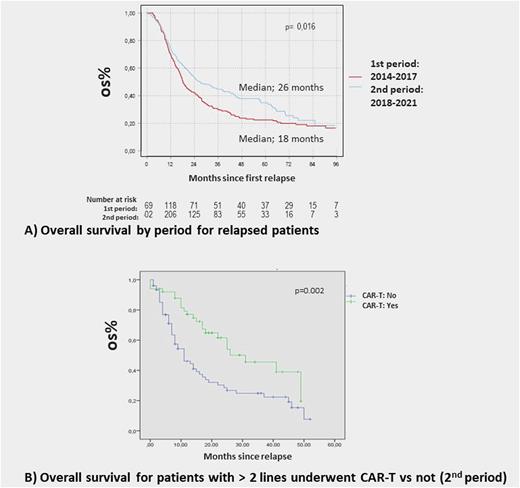
Five-year progression free survival (PFS) for the entire cohort was 71% (95% CI 69-74), and median overall survival (OS) was 90 months (95% CI: 76-NR). Median OS for r/r patients was 22 months (95% CI: 19 - 26), and for the group of patients who received > 2 lines was 23 months (95% CI; 19-27). With a median follow up of 25 months (50 months for the 1st period and 20 months for the 2nd period) OS was superior for patients relapsed in the 2nd period (26 months [95% CI: 22-38]), compared with those relapsed in the 1st period (18 months [IC: 16-22], p= 0.016) (Figure 1, A). Median OS since first relapse for patients pre-treated with > 2 lines who received CAR-T cells was 31 months vs 11 months for patients who did not receive CART (p= 0.002) (Figure 1, B).
Conclusion: The OS of patients with r/r DLBCL and HGBCL has improved in recent years, probably in relation to the incorporation of NT, although the use of these NT in Spain has been lower than expected for this population.
Disclosures
Bastos-Oreiro:JANSSEN: Speakers Bureau; Roche: Consultancy, Research Funding, Speakers Bureau; KITE/GILEAD: Consultancy, Honoraria; NOVARTIS: Speakers Bureau; INCYTE: Consultancy, Speakers Bureau. Jimenez-Ubieto:Novartis: Consultancy. González de Villambrosia:EusaPharma: Honoraria; Incyte: Honoraria; Janssen: Honoraria; Takeda: Honoraria. Cordoba:GenMab: Consultancy; Pfizer: Consultancy, Speakers Bureau; Kite: Consultancy; Takeda: Consultancy; Bristol Myers: Research Funding; Celgene: Consultancy, Honoraria; Gilead: Honoraria. Pérez Ceballos:Takeda: Consultancy, Honoraria; Janssen: Consultancy; Incyte: Consultancy; Roche: Honoraria. Muntañola Prat:Roche: Honoraria, Speakers Bureau; Abbvie: Honoraria, Speakers Bureau; AstraZeneca: Honoraria, Speakers Bureau; Janssen: Honoraria, Speakers Bureau. Martín García-Sancho:Incyte: Consultancy, Other: Support for attending meetings and/or travel; Lilly: Consultancy, Other: Support for attending meetings and/or travel; Takeda: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; ADC Therapeutics America: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Miltenyi: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Kern: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Servier: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Gilead/Kite: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Novartis: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Eusa Pharma: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Clinigen: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Kyowa Kirin: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Bristol Myers Squibb/Celgene: Consultancy, Honoraria, Other: Support for attending meetings and/or travel; Roche: Consultancy, Honoraria, Other: Support for attending meetings and/or travel.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal